Textbook Question
(b) Without looking at a periodic table, list as many alkaline earth metals as you can. (There are five common ones.)
445
views

 Verified step by step guidance
Verified step by step guidance

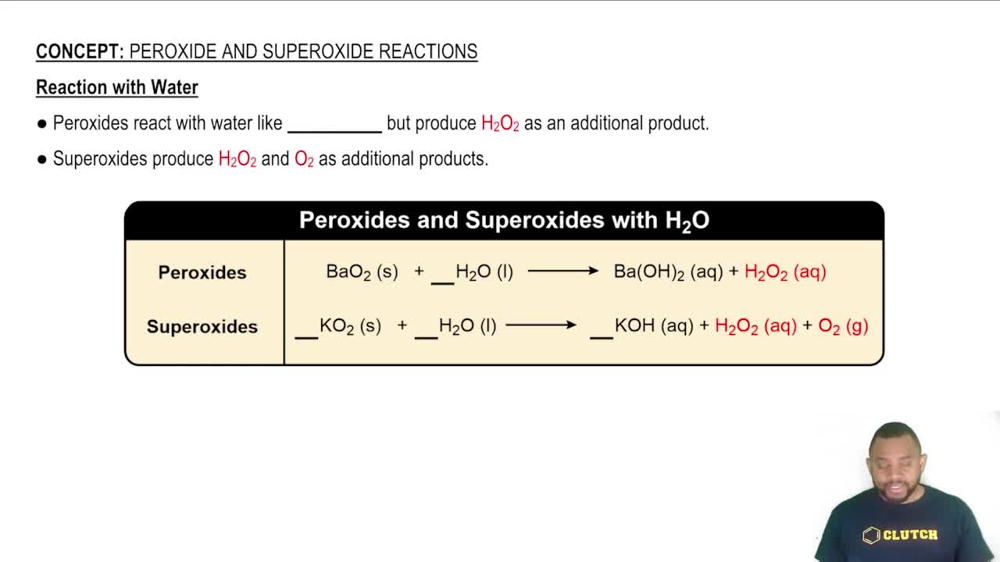
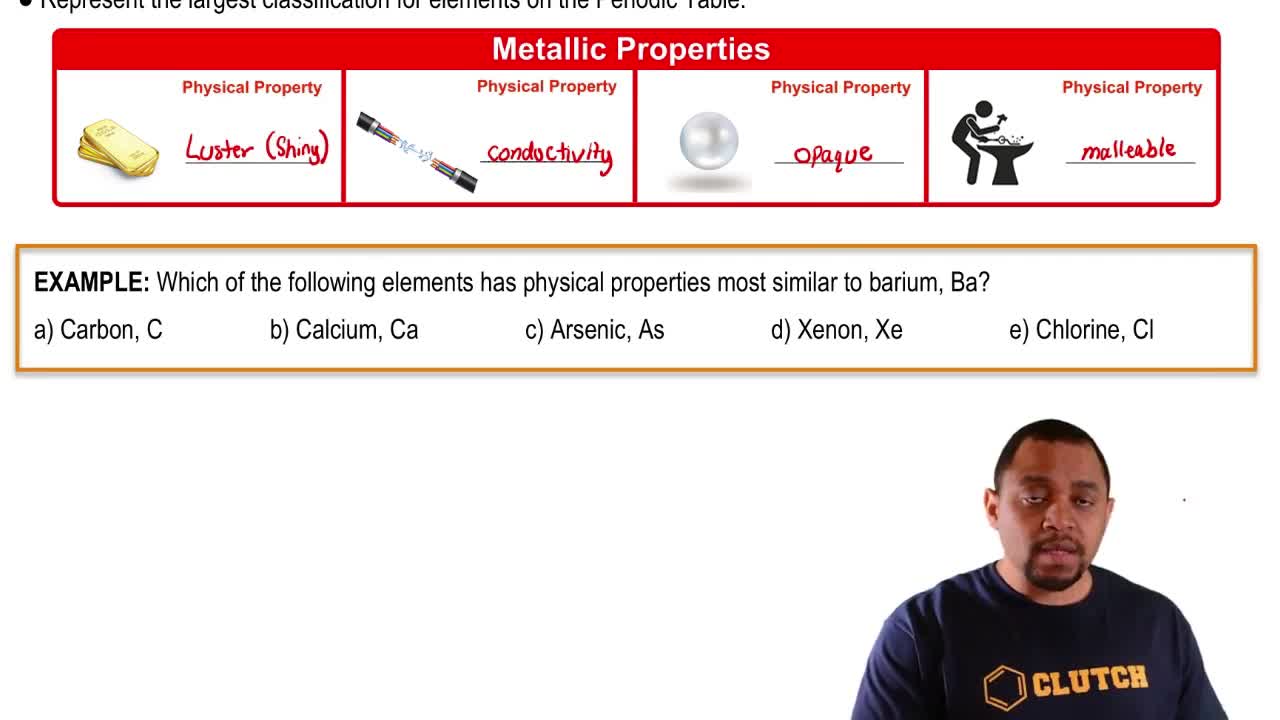
(b) Without looking at a periodic table, list as many alkaline earth metals as you can. (There are five common ones.)