Textbook Question
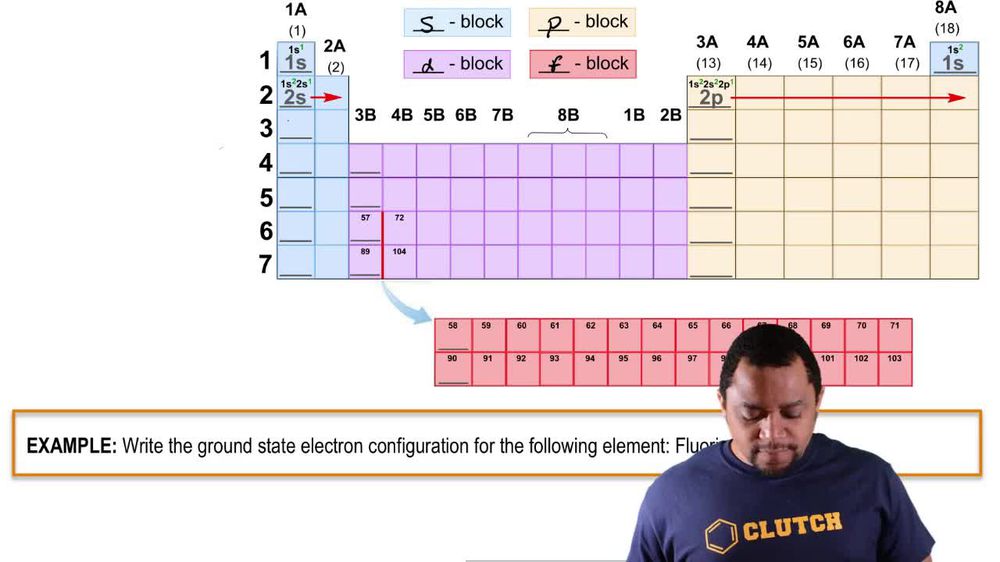
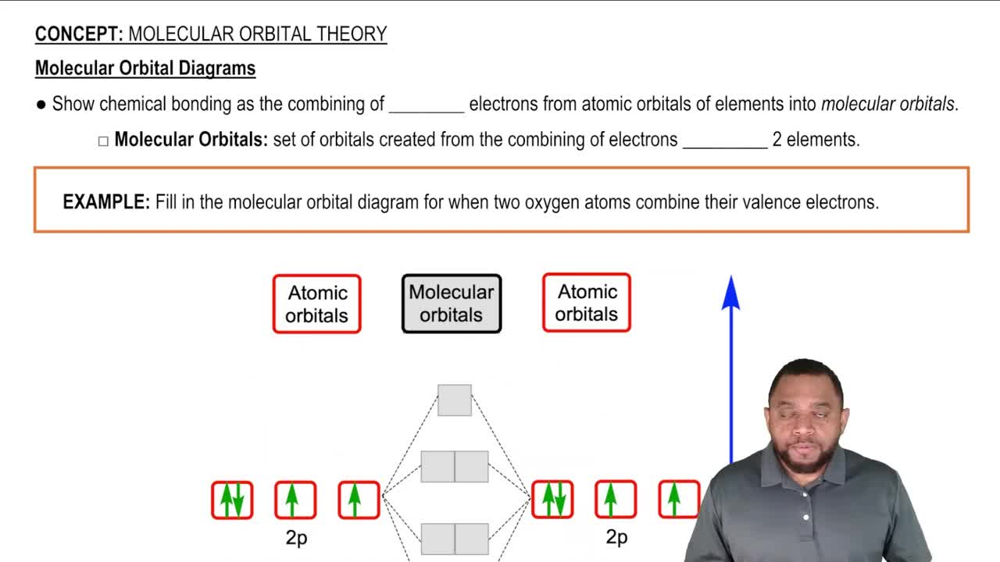
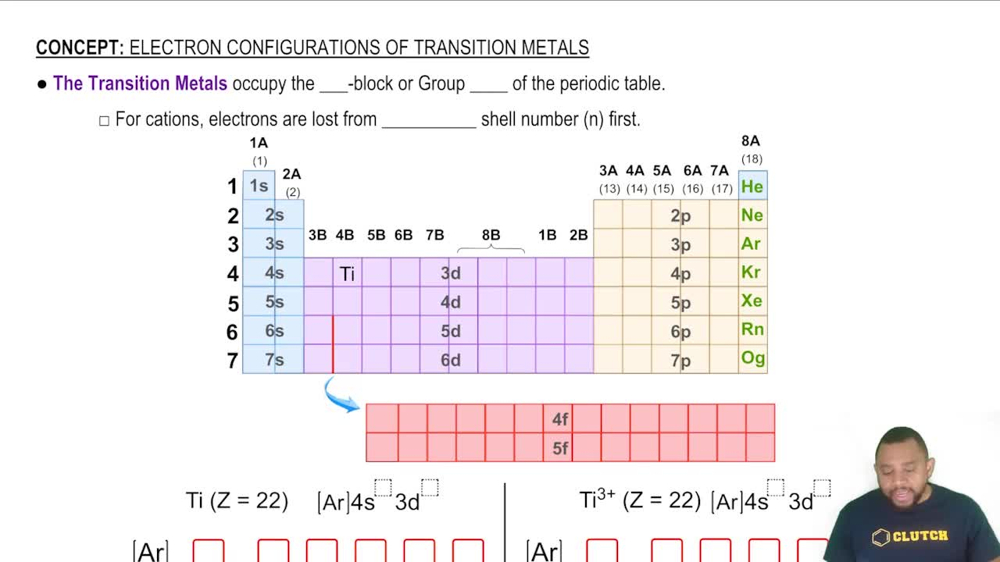
Draw orbital-filling diagrams for atoms with the following atomic numbers. Show each electron as an up or down arrow, and use the abbreviation of the preceding noble gas to represent inner-shell electrons. (a) Z = 25 (b) Z = 56 (c) Z = 28 (d) Z = 47
902
views