Textbook Question
Which outer electron configurations would you expect to belong to a noble gas? a. ns2 b. ns2np6 c. ns2np5 d. ns2np2
975
views

 Verified step by step guidance
Verified step by step guidance


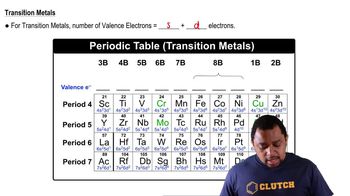
Which outer electron configurations would you expect to belong to a noble gas? a. ns2 b. ns2np6 c. ns2np5 d. ns2np2
Determine the number of valence electrons in an atom of each element. a. Ba c. Ni d. S
Which outer electron configuration would you expect to belong to a reactive nonmetal? a. ns2 b. ns2np6 c. ns2np5 d. ns2np2
Which outer electron configurations would you expect to belong to a metalloid? ns2 b. ns2np6 c. ns2np5 d. ns2np2
Determine the number of valence electrons in each element. Which elements do you expect to lose electrons in chemical reactions? Which do you expect to gain electrons? a. Al b. Sn c. Br d. Se
Determine the number of valence electrons in an atom of each element. b. Cs