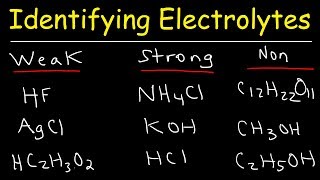
6. Chemical Quantities & Aqueous Reactions
Electrolytes
Problem 65
Textbook Question
Textbook QuestionA solution of HCl in water conducts electricity, but a solu-tion of HCl in chloroform, CHCl3, does not. What does this observation tell you about how HCl exists in water and how it exists in chloroform?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
1071
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 14 videos