Textbook Question
For each element, indicate the number of valence electrons, core electrons, and unpaired electrons in the ground state: (c) fluorine.
563
views
1
comments

 Verified step by step guidance
Verified step by step guidance
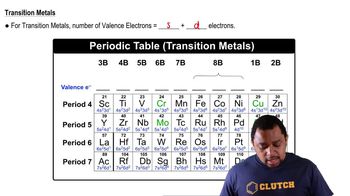


For each element, indicate the number of valence electrons, core electrons, and unpaired electrons in the ground state: (c) fluorine.
For each element, indicate the number of valence electrons, core electrons, and unpaired electrons in the ground state: (a) sodium (b) sulfur.
What is the maximum number of electrons that can occupy each of the following subshells? (a) 3s, (b) 2p, (c) 4d, (d) 5s.
(c) What does each box in an orbital diagram represent?