11. Bonding & Molecular Structure
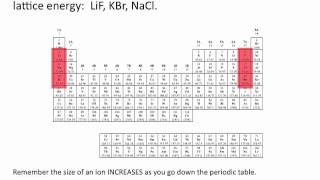
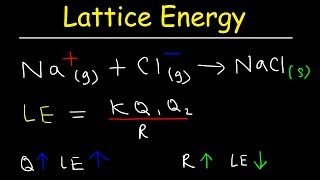
Lattice Energy

Get help from an AI Tutor
Ask a question to get started.
Problem 22a
Textbook Question
Textbook QuestionNaCl and KF have the same crystal structure. The only difference between the two is the distance that separates cations and anions. (b) Use the ionic radii given in Figure 7.8 to estimate the Na¬Cl and K¬F distances.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
387
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 13 videos