11. Bonding & Molecular Structure
Chemical Bonds
Problem 41
Textbook Question
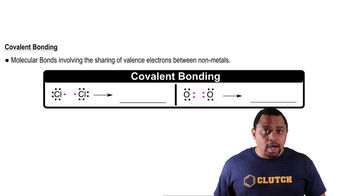
Textbook QuestionWhy do two atoms come together to form a covalent bond? (a) Attractive forces between the positively charged nuclei and the electrons in both atoms occur when the atoms are close together. (b) Repulsive forces between protons in the nuclei of the two atoms are minimized when the atoms are close together. (c) Repulsive forces between electrons in the two atoms are minimized when the atoms are close together.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
935
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 14 videos