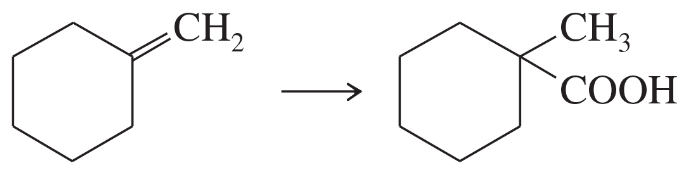
Show how the following compounds could be prepared from the given starting materials. You can use any necessary organic or inorganic reagents.
c.
 Verified step by step guidance
Verified step by step guidance Verified video answer for a similar problem:
Verified video answer for a similar problem:



 2:12m
2:12mMaster Carbonation of Grignard Reagents with a bite sized video explanation from Johnny
Start learning