Draw the following sugars using Haworth projections:
b. α-D-tagatopyranose
 Verified step by step guidance
Verified step by step guidance Verified video answer for a similar problem:
Verified video answer for a similar problem: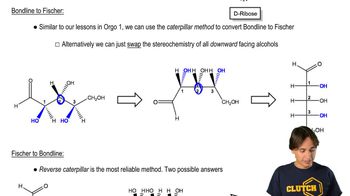

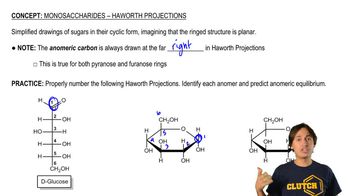

 4:03m
4:03mMaster Monosaccharides - Haworth Projections with a bite sized video explanation from Johnny
Start learning