Use resonance structures to identify the areas of high and low electron density in the following compounds:
(a)
(b)
(c)


 Verified step by step guidance
Verified step by step guidance Verified video answer for a similar problem:
Verified video answer for a similar problem:
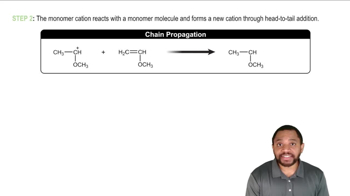


 3:34m
3:34mMaster The rules you need for resonance: with a bite sized video explanation from Johnny
Start learning