Open Question
Beginning from Benzene, synthesize the following compound.
12
comments
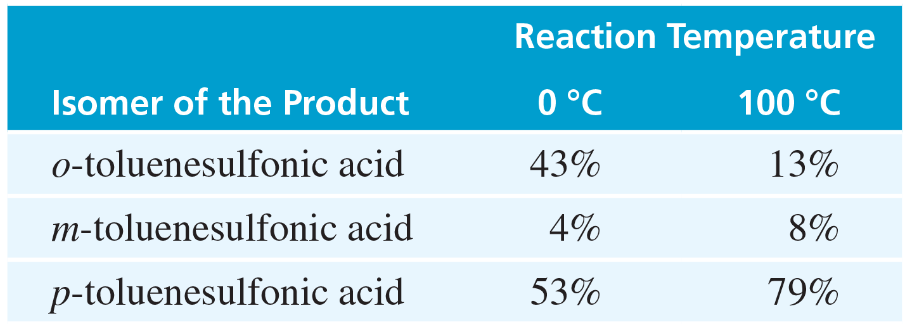
 Verified step by step guidance
Verified step by step guidance Verified video answer for a similar problem:
Verified video answer for a similar problem:



 4:33m
4:33mMaster Forcing Ortho substitution with a bite sized video explanation from Johnny
Start learning