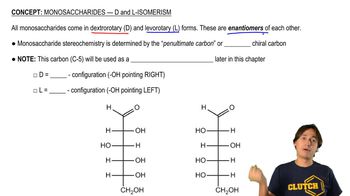
Textbook Question
a. Draw a Lewis structure for each of the following:
b. Draw a structure for each of the species that shows approximate bond angles.
c. Which species have no dipole moment?
3. N3−
1279
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 1 - Remembering General Chemistry: Electronic Structure and Bonding (Part 2)
Ch. 1 - Remembering General Chemistry: Electronic Structure and Bonding (Part 2) Problem 78
Problem 78 Verified step by step guidance
Verified step by step guidance

a. Draw a Lewis structure for each of the following:
b. Draw a structure for each of the species that shows approximate bond angles.
c. Which species have no dipole moment?
3. N3−
Draw a Lewis structure for each of the following:
4. CH3CONH2
b. Draw a structure for each of the species that shows approximate bond angles.
c. Which species have no dipole moment?