Predict the products of the following acid–base reactions, or indicate if no significant reaction would take place.
a. H—C≡C—H + NaNH2
b. H—C≡C—H + CH3Li
c. H—C≡C—H + NaOCH3
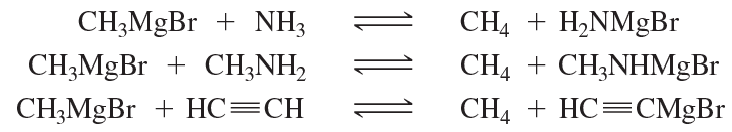
 Verified step by step guidance
Verified step by step guidance Verified video answer for a similar problem:
Verified video answer for a similar problem:


 5:11m
5:11mMaster The 3 steps for determining the direction of acid and base equilibrium. with a bite sized video explanation from Johnny
Start learning